Explore our latest innovations in leak testing of transfer systems and glove integrity testing at INTERPHEX 2026, April 21–23 in New York.
Visit us at Booth 2662 to discover technologies designed to strengthen contamination control and enhance the reliability of aseptic manufacturing.
As regulatory expectations continue to evolve, pharmaceutical manufacturers face increasing pressure to ensure the integrity of barrier systems. Our solutions provide the precision, efficiency, and digital traceability needed to support a robust Contamination Control Strategy (CCS) and maintain consistent product quality.

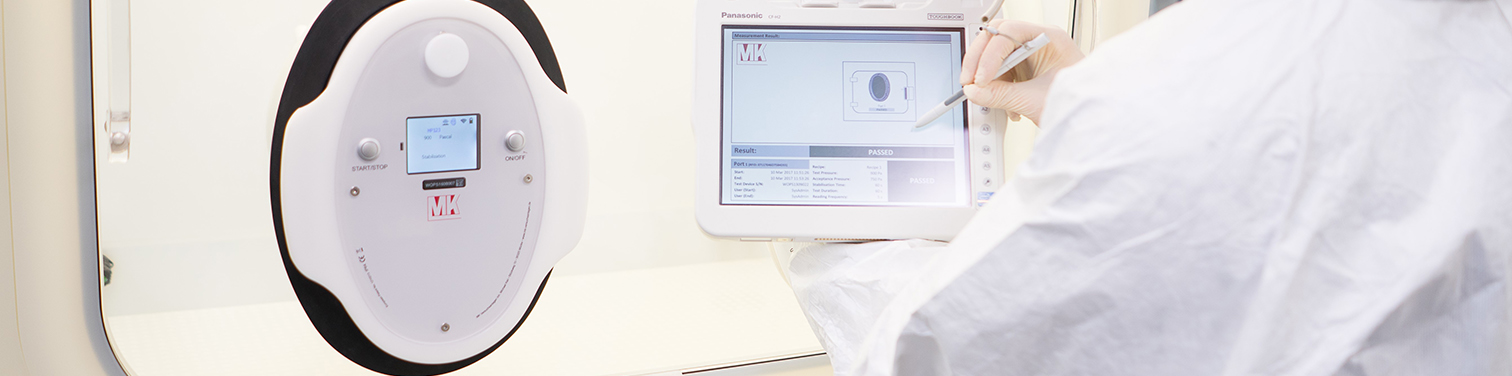
RTP Tester – closing a critical risk gap in aseptic processing
While glove integrity testing is already a mandatory requirement under Annex 1, there is currently no such strict regulatory obligation to test transfer ports or containers; however, due to current regulatory discussions, it is expected that this will become increasingly strict, and the industry is already preparing for this. At the same time, the contamination risk associated with these interfaces is equally significant. Every transfer step represents a potential breach point, and regulators are increasingly emphasizing holistic contamination control across all barrier components.
The RTP Tester enables you to address this risk proactively. With highly sensitive pressure decay testing, even the smallest leaks in alpha ports, beta containers, and other transfer system seals are detected within minutes, helping mitigate a contamination incident before it happens. This strengthens your contamination control strategy today and prepares you for future regulatory expectations.
Highlights include:
- Compatibility with alpha ports, beta containers & other transfer systems
- Individually configured test parameters for maximum accuracy
- Flexible sealing units adaptable to different formats
- Wi-Fi & RFID for seamless digital workflows
- MES integration and encrypted communication (WPA2)
- Smart container management and comprehensive service options
Glove integrity testing – automated, fast and fully compliant
Our Glove Integrity Testing Software (GITS®) sets a new benchmark in automated glove testing for isolators and RABS. It enables decentralized, simultaneous testing of all gloves in a system, significantly reducing downtime and supporting efficient production planning.
Key benefits include:
- Integrity testing from as little as 5 minutes
- GITS®4 software compliant with GAMP 5, FDA 21 CFR Part 11 & EU Annex 11
- RFID based glove and port recognition
- Full traceability, automatic reporting & lifecycle monitoring (GLCS®)
- Wireless data transfer and database-supported documentation
- Easy to clean, H₂O₂/VHP-compatible, and suitable for nearly all port sizes
With GITS®, you can reliably meet Annex 1 requirements for routine glove integrity testing — at the beginning and end of each batch or at defined intervals within your CCS.
Meet us at Booth 2662
Don’t miss the opportunity to discover our latest technologies and discuss how we can support your current projects and future goals.
Book your meeting here or request a free ticket code: INTERPHEX | MK METALFREE