The global vaccine market has seen significant growth due to the growing global population, increased immunisation coverage and new vaccines being introduced. With COVID-19 this trend is expected to accelerate. The billions of doses of COVID-19 vaccine required on top of the existing vaccine demand is just one of the dynamics influencing capacity planning for manufacturers. New treatments and more approvals every year, in particular biological drugs, also add to the global capacity demand.
To manage the situation, pharmaceutical companies continuously assess options for balancing available capacity by adding more capacity. For the short term, they can increase outsourcing to Contract Manufacturing Organisations (CMOs) or repurpose existing facilities. Looking beyond the short term, they can alternatively plan to build new facilities.
Large open “ballrooms” will provide flexibility and simplify reconfiguration
Availability of engineering capacity and expertise in planning, designing, and executing the projects are some of the challenges when establishing these complex facilities. Another challenge is how to ensure that the facility you decide to build will be flexible enough and can be adapted to future changes in the product mix. Time to market and ensuring the return of investment are also key for a successful capital investment project.
Vaccine companies, like pharmaceutical manufacturers, desire innovative facility solutions to address these challenges. To this end, offsite construction and modular facilities can be an attractive solution.
Facility design
When designing the next generation of vaccine manufacturing facilities, the following questions must be addressed:
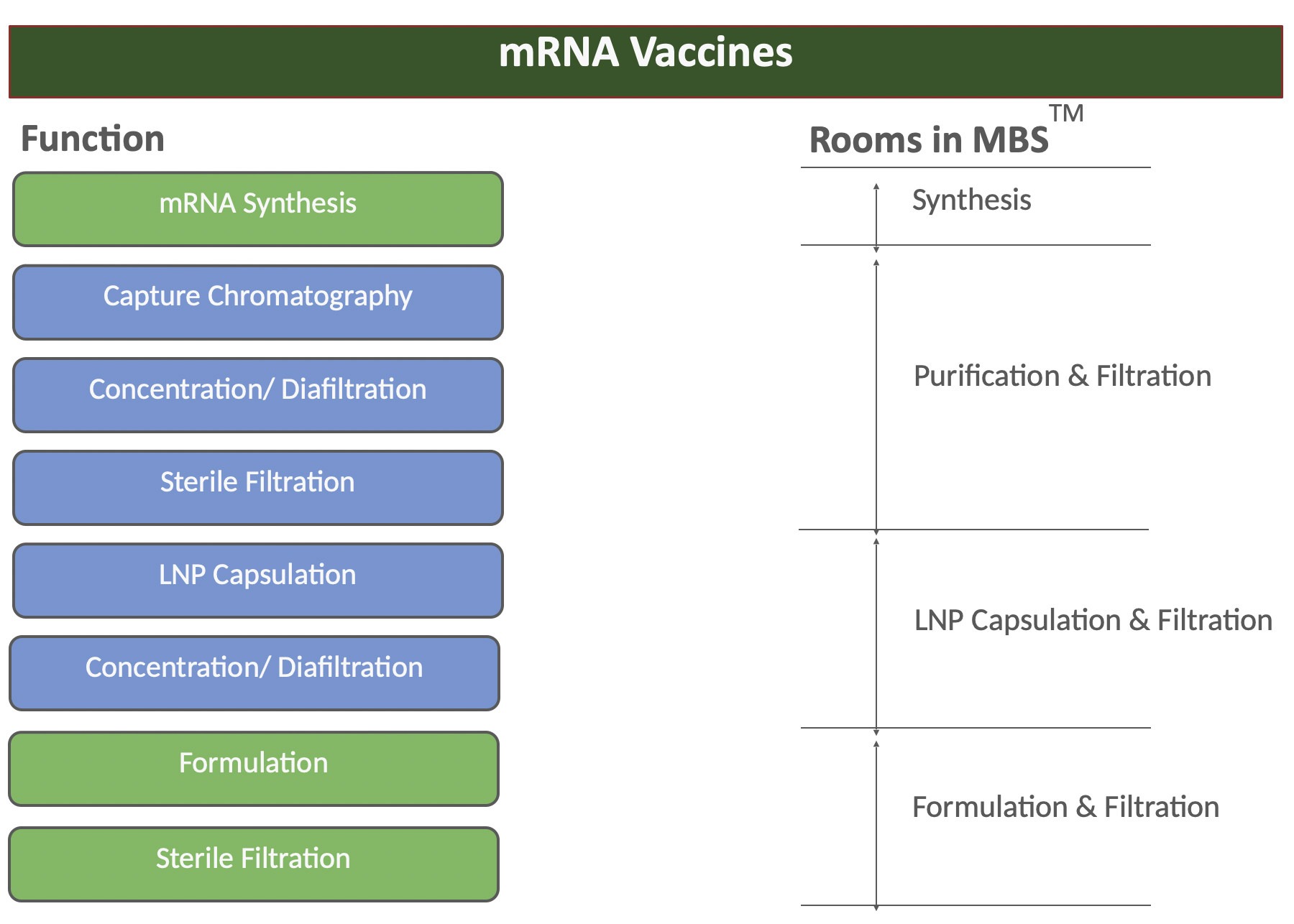
The key principle is to develop the layout with the manufacturing process as a starting point
- Which production environment, such as air turnover and containment, is required per unit operation of the vaccine manufacturing process?
- Which is the best strategy for separating the unit operations and what level of segregation is required for the different products to manufactured?
- What level of automation should be included for facility and process operations?
- Where will the facility be placed and are there any local requirements, space limitations or existing buildings that need to be taken into consideration for the design?
- Will the facility design enable reliable production and does the facility design enable meeting future regulatory expectations?
- Do the facility design and construction support rapid deployment and minimise the time to market?
- How can risks be avoided and how can safety be increased?
To meet these market expectations and to simplify the design and construction of turnkey facilities for vaccine and biomanufacturing, KeyPlants developed a pre-engineered design platform called Modular Bio Solutions, MBS and what was learnt from this experience is invaluable. Preparing a solution such as this requires a lot of planning to hit the right balance of flexibility and ease-of-use from standard layouts.
Next-generation facilities
When looking to build a modern-day vaccine facility solution, there are some important parts to consider for the standard layout. These include areas for material dispensing, media and buffer preparation and storage, upstream and downstream processing as well as sufficient space for all support functions, airlocks, and corridors. Clean utilities, HVAC, cleanrooms, processing equipment and building management systems are all part of this turnkey solution.
The facility layout must also be optimised to minimise the required footprint, resulting in reduced capital and lower operational costs.
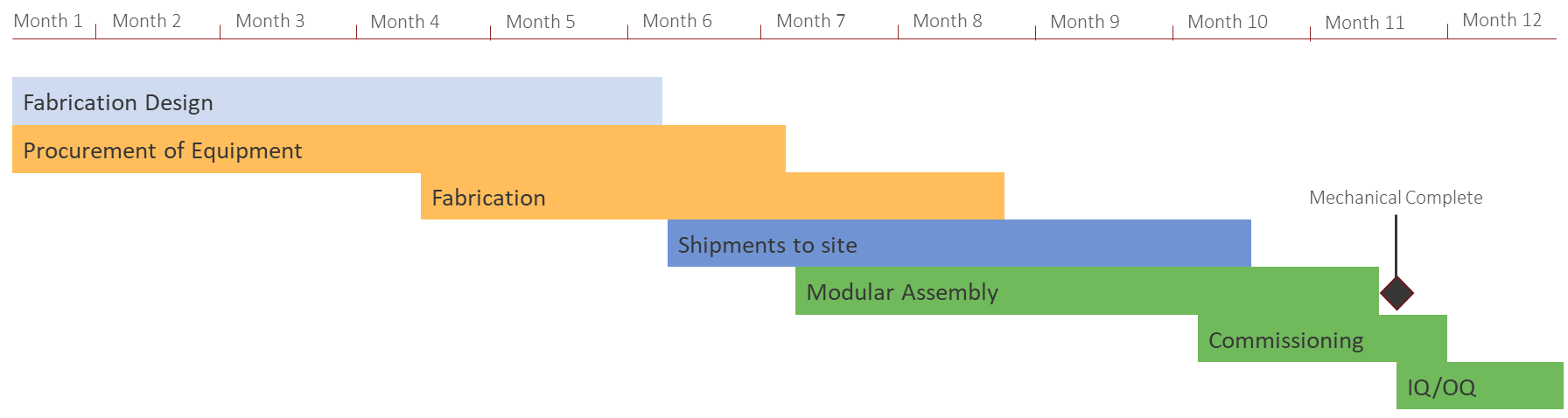
Figure 1: Facility timeline example based on Modular Bio Solutions MBS and disposables for bioprocessing
To build a standardised facility design that can be adapted to the required manufacturing technology and capacity it is important to establish a flexible layout that can be modified easily. For example, the capacity achievable with the standard design is scalable from 500 L up to multiple 2000 L bioreactors. The level of segregation is set according to product and regulatory requirements and flexible cleanroom panels are used for simple layout configuration.
Other operations such as formulation, aseptic filling, and lyophilisation are typically performed in a separate facility that can be placed adjacent to the bulk manufacturing facility.
A modular vaccine manufacturing facility can be constructed and commissioned in less than 12 months, for deployment anywhere in the world. A typical timeline for design, procurement, fabrication, transport, assembly and commissioning of such facility is shown in Figure 1.
Offsite construction and modular facilities can be an attractive solution to reduce time to market
The key principle when designing a facility is to develop the layout with the manufacturing process as a starting point. When the process steps are set, the required manufacturing rooms can be identified. Table 1 illustrates the design principle for an mRNA vaccine as an example, explains the categories each step of the process fits within. With the functions and process rooms set, an adjacency study is typically performed to optimise the layout, and finally, the complete facility can be detailed.
Depending on which vaccine is to be manufactured in the facility, there will be differences in required process steps, space per function and segregation to mention a few. Ultimately the required footprint and complexity will differ significantly depending on the process and capacity.
Look to the future
It is looking likely that the way engineering and capital projects are managed will change. Reducing timelines is one of the major change drivers in capital projects for vaccine and pharmaceutical manufacturers. Offsite construction and modular solutions enable manufacturers to dramatically reduce their timelines and meet their objectives.
More new drugs approvals, additional indications, more biologics, and more niche products have created a demand for flexible facilities for biomanufacturing in general, including vaccines. New facilities need to be reconfigurable to meet future changes in volumes and product mix.
Large open spaces, "ballrooms", will provide flexibility and simplify reconfiguration. Walls in the modular building that can be moved, and room classification that can be changed are other examples of reconfigurability. Modular facilities, designed by an experienced team, will have this flexibility and can easily be expanded and even moved.
In conventional capital investment projects, delays, orders changes and rework are common challenges. There is a strong demand for increased predictability with regards to time, quality, and costs. Offsite construction with fabrication in a workshop will provide this predictability as 80% of the work is done in a controlled environment.
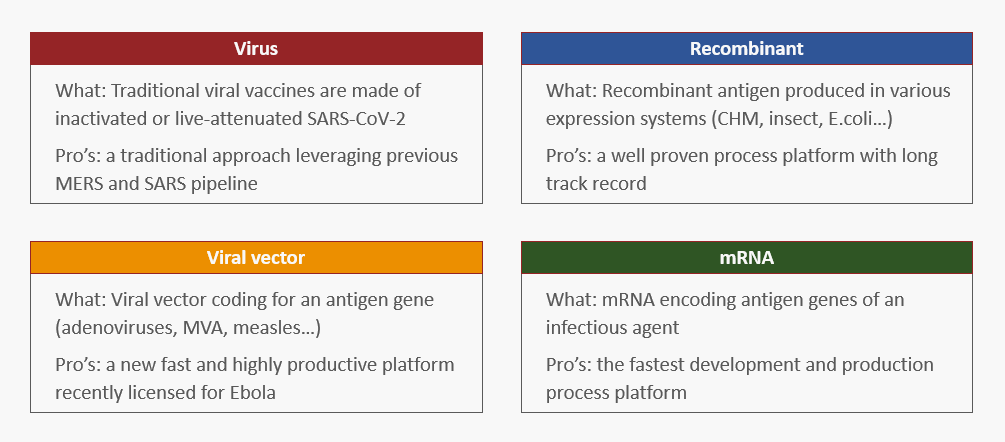
In summary, there is clearly a market demand to establish new vaccine facilities with modular solutions providing speed, flexibility, and predictability. As the pandemic truly began at the beginning of 2020, our front-end design team immediately developed our concepts to support what would likely be needed for efficient COVID-19 vaccine manufacturing. Based on our design platform for cell culture manufacturing, pre-engineered COVID-19 vaccine facilities were developed. As a result, design platforms and facilities for manufacturing of virus-, recombinant-, viral vector- and mRNA-based vaccines are now available from KeyPlants.




