Manufacturing specialised medicines in a pharmaceutical production environment has a specific set of requirements when it comes to hand protection. Production and handling of these types of products typically happen in a cleanroom or controlled environment and quite often in a sterile environment as well.
When this application is specialised even further into manufacturing or handling of a particular set of pharmaceuticals known as chemotherapeutic agents, additional requirements come into play when selecting appropriate hand protection. Examples of these types of drug products and the appropriate protective glove are listed in the table below.
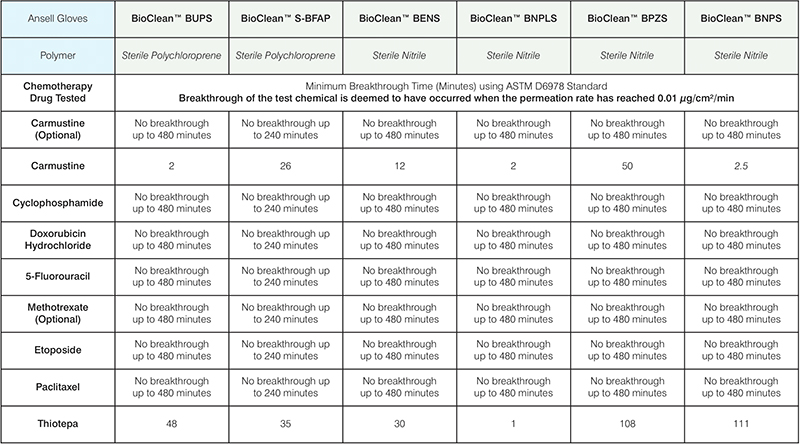
For sterile and cleanroom environments the following products
are recommended along with their breakthrough time
There are two primary reasons to wear personal protective gloves when working with these types of drugs. First and foremost to protect the individual from exposure to a potentially harmful substance, and secondarily to protect the product from contamination.
Handled with care
Chemotherapeutic agents are a class of chemical compounds designed and formulated as a drug product to inhibit the growth of or destroy rapidly growing cancer cells within the body. By definition, they are either cytostatic or cytotoxic and as such require the use of personal protective gloves that will act as an effective barrier between the hand and the chemical compound in question. Since these compounds are by nature destructive to human cells it is desirable to avoid exposure to these compounds.
How do individuals potentially exposed to these types of chemical compounds, know whether or not the gloves they are wearing will provide adequate protection? Gloves designed to be used in these drug-making environments can be evaluated for their protective qualities when in contact with chemical substances. The evaluation is done by conducting a chemical permeation test under the guidance of two US industry consensus standards known as “ASTM D6978
Assessment of Resistance of Medical Gloves to Permeation by Chemotherapy Drugs” and “ASTM F739 Standard Test Method for Permeation of Liquids and Gases through Protective Clothing Materials under Conditions of Continuous Contact”.
While ASTM F739 is the general test method used to conduct chemical permeation testing, the ASTM D6978 includes some additional requirements specific to chemotherapy drugs.
The ASTM F739 standard test method is used to identify the actual chemical permeation resistance of glove materials under continuous contact with chemicals. The glove material to be tested is placed into a permeation test cell and sandwiched between the test chemical and a collection medium. The collection medium, usually a gas or liquid, is analysed quantitatively for its concentration of the chemical that has permeated the barrier as a function of time after its initial contact with the glove material.
Each material specimen to be tested is sampled from the palm of at least three gloves. An additional sample may be tested with just collection media as a test control depending upon the actual analytical methods used. All test specimens are cut to fit the same diameter as the flange of the permeation test cell.
Permeation: process by which a chemical dissolves and/or moves through a protective glove material on a molecular level
The test chemical is introduced into the challenge compartment of the permeation cell and the time measuring device is started. The compartment containing the test chemical is completely filled during the period of the test. Under the requirements of ASTM F739, the breakthrough time of a chemical is deemed to occur when the sum of the permeation rates of each individual component reaches the rate of 0.1µg.cm2/m.
When permeation rate of 0.1µg.cm2/m is detected, then the breakthrough time is reported in minutes for each test specimen. If the permeation rate does not reach 0.1µg.cm2/m, then the duration of the test is reported.
For chemotherapy agents under the additional requirements of the standard ASTM D6978, a more conservative breakthrough time is reported by determining a breakthrough time when 0.1µg.cm2/m is reached. This is done in recognition of the cytotoxic/cytostatic properties of the chemical compounds in question.
Ansell gloves are tested against the most stringent standard, the American ASTM D6978- 05, which employs a testing limit 100 times more stringent than its European counterpart. We do not test gloves using the EN16523-1:2015 (formerly EN374-3) method as this benchmark is not safe when assessing the suitability of a glove for protection against chemotherapy drugs.
Penetration: the movement of a chemical and/or microorganism through the material, pinholes or other imperfections of a glove
To illustrate how the two standards parameters compare, we have highlighted the consequences in the table below.
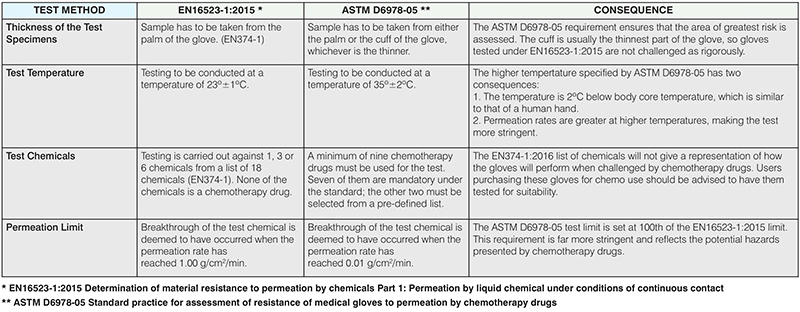
* EN16523-1:2015 Determination of material resistance to permeation by chemicals Part 1: Permeation by liquid chemical under conditions of continuous contact
** ASTM D6978-05 Standard practice for assessment of resistance of medical gloves
to permeation by chemotherapy drugs
Contamination control on the product
While personal protection is the first concern when selecting a glove, protecting the product from external sources of contamination is equally important: manufacturing of chemotherapy drugs is conducted under good manufacturing practices (GMP) in a sterile cleanroom environment.A variety of sources of potential contamination must be taken into consideration, including biological, particulate and undesirable chemical residues. A contaminated product from any of these sources can lead to unacceptable production lots resulting in a costly and time- consuming scenario to rectify.
Factors need to be taken into consideration when choosing an appropriate glove chosen for use with chemotherapy agents include:
- Protection against specific drugs being used
- and hazards or chemicals in the workplace
- Protection of products from external contamination
- Comfort
- Fit
- Ergonomics
- Costs
Additionally, a common practice of wearing two pairs of single-use gloves (double donning) can also enhance the end user's protection against chemotherapy agents provided the gloves are chemotherapy drug approved and proven to be elastic and comfortable.
N.B. This article is featured in the March 2020 issue of Cleanroom Technology. Subscribe today and get your print copy!
The latest digital edition is available online.





